Consumers
Single-celled primary producers in the ice (autotrophic algae and bacteria) form the basis of the food web in the ice: they absorb CO2 and, drawing on the energy of light, use it to produce carbon compounds (photosynthesis). They are referred to as being ‘self-sustaining’, that is, as autotrophic or phototrophic. In contrast, other organisms living in the sea ice take up carbon through their food; they are heterotrophic. These include single-celled organisms, the protists or protozoa, which feed on algae, but also on bacteria and archaea (formerly classified as bacteria). In this regard, the most common group are the ciliates. Flagellates are also common, including heterotrophic dinoflagellates. These also include the so-called kleptoplastic dinoflagellates, which are capable of preserving the chloroplasts from the algae or bacteria they consume, and subsequently using them for photosynthesis. Other flagellates in the sea ice include the choanoflagellates. Other heterotrophic protists include gymnamoebia, foraminifera, acantharians and radiolarians (Caron et al., 2017), which can extend parts of their body to capture prey. Today, the microbial food webs in the sea ice remain poorly understood. However, recent studies indicate that heterotrophic protists employ a wide range of feeding strategies.
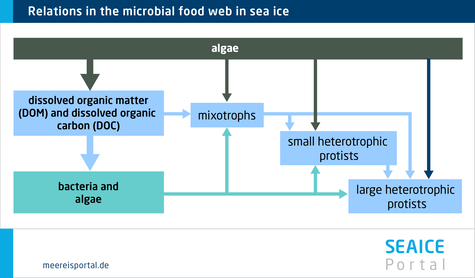

Just like algae, bacteria and archaea, the single-celled consumers (protozoa) also find their way into the sea ice while it’s forming. They are, in a sense, encased in the ice structure. The majority can be found in brine channels, in pressure ridges, in meltwater at the snow-ice interface, on the under-side of the ice, and in meltwater pools on the Arctic sea ice (Caron et. al., 2017). These hetero-trophic protists live under extreme conditions: the salinity varies greatly between the brine channels and the seawater (in the Arctic Ocean the salinity level is circa 32; in the brine channels, it can range from 42 to 93 (Wakatsuchi and Ono, 1983) . The temperature in the ice can vary signifi-cantly, and the light intensity not only varies throughout the annual cycle, but also differs between e.g. the surface and underside of the ice. Although low temperatures generally result in declining feeding behaviour and growth rates, the effect of temperature fluctuations on growth is still only partially understood.
Heterotrophic protists can above all be found in places where there are ample bacteria and algae for them to feed on. Their population reaches maximum growth in summer, which is when the available food is at its highest. Accordingly, the greatest biomass is to be expected when the ice melts in late summer. During this time, the heterotrophic protists’ biomass and waste products enter the seawater as particles, making a substantial contribution to the input of organic material in the water column.For example, in the coastal areas of the Antarctic, single-celled consumers (protozoa) from the sea ice account for nearly 20 percent of total integrated microbial biomass. Up to ten percent of the biomass attached to the bottom of the sea ice (Caron et al., 2017). In Greenland’s pack ice, heterotrophic flagellates alone account for 20 percent of total microbiological biomass on average. The relative distribution of autotrophic and heterotrophic protist species is similar in the Arctic and Antarctic. Their absolute abundance in sea ice (number of cells per given volume of melted sea ice) is often one to two orders of magnitude higher than in seawater (number of cells per given volume of seawater).
Micro-consumers in sea ice ...
- consume particulate organic carbon (e.g. EPSs, bacteria or algae) and fulfil an important role in biogeochemical cycles in the polar regions.
- remineralise the organic compounds they consume, producing inorganic nutrients. These serve as a substrate for bacteria to grow on; in turn, the bacteria drive primary production.
- are an important component in food relations (trophic interactions) within the marine organic community.
- form, together with ice algae, a reserve of organic material, which becomes encased in the ice in winter, offering both a food source for overwintering organisms and a ‘kick-start’ for the ecosystem in spring.
Caron, D. A., R. J. Gast & M.-E. Garneau (2017): Sea ice as a habitat for micrograzers, In: Sea Ice, D. N. Thomas (ed.), 3rd edition, Wiley-Blackwell, Chichester (UK), Hoboken (NJ), 370-393.
Lizotte, M.P. (2003): The Microbiology of Sea Ice, In: D. N. Thomas & G.S. Dieckmann (eds.) Sea Ice, Oxford: Wiley-Blackwell, 201.
Wakatsuchi M. & N. Ono (1983): Measurements of salinity and volume of brine excluded from growing sea ice, Journal of Geophysical Research Oceans, doi.org/10.1029/JC088iC05p02943
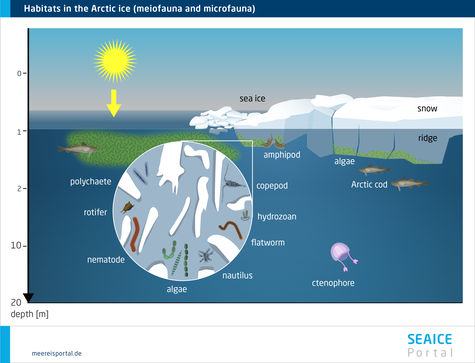
In this chapter we will describe the metazoa (multicellular fauna) associated with sea ice. They include both the small organisms inhabiting the ice’s brine-channel system (meiofauna) and the larger animals that can be found in the ice-water interface layer (under-ice fauna) (Bluhm et al., 2017).
The majority of species in the ice can also be found in the water column and / or on the seafloor; very few exclusively inhabit the sea ice. Though some taxa can be found in both polar regions, rotifers (wheel animals) and nematodes are more common in the Arctic sea ice, while copepods are more often found in the Antarctic sea ice. To date, cnidarians and ribbon worms (nemerteans) have so far only been found in Arctic sea ice, whereas comb jellies (ctenophores) and sea slugs (nudibranchs) have only been found in the brine-channel systems of Antarctic pack ice. The diversity of the under-ice metazoans depends on the extent to which the upper water column is included. Generally speaking, the biodiversity there is higher than in the ice; at least 40 species have been confirmed (Bluhm et al., 2017). These include crustaceans like copepods and krill, fish and turbellarians. In addition, the free-swimming larvae of fish and crabs can be found here (Arrigo, 2014 ). The animals use the sea ice for grazing, or as protection from predators, forming a complex food web.
Given its major seasonal and regional variations, sea ice offers a range of habitats for fauna that have adapted to it. In the course of the year, the sea ice changes and with it, the living conditions for the fauna. Population sizes fluctuate throughout the annual cycle, rising substantially in spring. This increase can constitute one to four orders of magnitude (Arrigo, 2014 ). Enormous numbers of individual organisms per square metre can be reached: from fewer than 10 to more than 300,000 in the brine channels, and from 0 to more than 10,000 below the ice (Bluhm et al. 2017). In this regard, a distinction is made between organisms that permanently or only temporarily use the sea ice as habitat. Some Arctic species spend their entire lifecycle in or below the sea ice. Others only come there during a specific life phase, or reproduce there, and are released back into the seawater when the ice melts. Others only come when the food concentration is high (Bluhm et al. 2017).
The meiofauna include small, often elongated multicellular organisms that are capable of living in the channel system within the ice, e.g. various wormlike organisms like nematodes and turbellarians, but also rotifers, copepods, hydroid polyps and slugs. They all form an intricate food web in which they are protected from larger predators (Werner, 2014). Some sympagic (= living in the sea ice) species (e.g. the Antarctic slug Tergipes antarctica and the copepod Stephos longipes) are extraordinarily well adapted to low temperatures and varying salinities, with unique ‘anti-freeze’ protection like thermal hysteresis and osmotic substances. Such genetic and physiological adaptations are a prerequisite for living in this extreme habitat in the long term (Werner, 2014).
The underside of the sea ice is a unique habitat, which is influenced by the characteristics of the ice, and of the water layer directly below it, alike. A critical factor for the colonisation of this interface layer: the morphology of the ice’s underside, which, with its cracks and crevices, ledges and protuberances, creates specific microhabitats, e.g. by changing the water flow speed. Since the biomass of the sympagic algae, protozoa and metazoa is most often concentrated in the bottom-most centimetres of the ice, the underside of the ice – except in winter, when new ice forms there – offers ample food (Werner, 2014).


In addition, the underside of the ice can – depending on the season – also be highly diverse. Organisms living under the ice are important bridge-builders between sea-ice algae production and the organisms in the higher trophic levels, for which they are a food source. In both polar regions there are large populations of various copepods, which use the ice-water interface layer as a permanent or temporary habitat for finding food, or as a nursery for their young stages (e.g. eggs, larvae), under the pack ice. Aside from its function as a habitat in its own right, the underside of the ice is important for a range of processes that connect the sea ice and pelagic (and even benthic) habitats via material and energy flows, e.g. the previously mentioned feeding behaviours and migrations of sympagic, pelagic and benthic species, as well as the entrapment and release of organisms during ice formation and melting (Winter, 2014).
In the Arctic, perhaps the most noticeable fauna are several species of under-ice amphipods, which hang ‘upside down’ from the underside of the ice, where they spend their entire, several-year-long lifecycle (Winter, 2014). Their unique ecological role consists in providing a trophic link between the sea ice and water column, since they are important prey species for cryopelagic fishes (e.g. the Arctic cod) and diving seabirds (e.g. the little auk) (Winter, 2014). In the Antarctic, in contrast, this role is above all filled by two species of krill (Euphausia superba, E. crystallorophias), which settle on the underside of the pack ice as macrofauna, although only in winter and spring, when they can use the ice-algae biomass as a food source. In the Southern Ocean, the Antarctic krill Euphausia superba is an important keystone species for the entire Antarctic ecosystem. The species can only be found in the Antarctic, and in certain regions of the Southern Ocean it is the dominant secondary consumer after the copepods. The krill and copepods live in the uppermost water layers and under the ice. Here they graze on the ice algae, particularly in winter and spring, when there haven’t yet been any substantial phytoplankton blooms.




In the polar food web, macro-consumers fulfil an important role. Macro-consumers …
- that live in the brine channel system within the sea ice consume particulate organic carbon (e.g. EPSs, bacteria or algae) and excrete inorganic nutrients.
- like sea-ice amphipods are relevant as food sources for the only two ice-associated fish species in Arctic waters: the Arctic cod and Greenland cod (Boreogadus saida, Arctogadus glacialis ).
- like the copepods and amphipods living under the ice are important bridge-builders be-tween sea-ice algae production and higher trophic levels.
- like the Antarctic krill (Euphausia superba) are crucial to the Antarctic ecosystem. Various Antarctic krill species fulfil key positions in the Antarctic food web.
Arrigo, K.R. (2014): Sea ice ecosystems, Annual Review of Marine Science, vol. 6, pp. 439-467.
Arrigo, K.R. (2017): Sea ice as a habitat for primary producers, In: D.N. Thomas (ed.) Sea Ice, 3rd edition, Wiley-Blackwell, Chichester (UK) Hoboken (NJ), 352-369.
Bluhm, B.A., K. M Swadling & R. Gradinger (2017): Sea ice as habitat for macrograzers, In: D.N. Thomas (ed.) Sea Ice, 3rd edition, Wiley-Blackwell, Chichester (UK) Hoboken (NJ), 394-414.
Schiel, S., Cornils, A., Niehoff, B. (2017). Leben im Pelagial. In: Hempel, G., Bischof, K., Hagen, W. (eds) Faszination Meeresforschung. Springer, Berlin, Heidelberg, 27-40.
Schnack-Schiel, Sigrid. (2010). Stephos longipes - ein Eiscopepode. Naturwissenschaftliche Rundschau. 63. 596-597.
Werner I. (2014): Das Meereis als Lebensraum, In: Lozán, J.L., H.Grassl, D.Notz & D.Piepenburg: WARNSIGNAL KLIMA: Die Polarregionen. Wissenschaftliche Auswertungen, Hamburg, 140-144.