Sea Ice for Beginners
Taking a ship on a jaunt across the open water and crossing the North Pole? It’s hardly imaginable. But a few decades from now, it might be a reality in the summer – since sea is rapidly declining due to climate change, especially in the Arctic.
For most of us, the frigid yet beautiful polar world, with its endless expanses, polar bears and walruses, is very, very far away. So why should we care about the sea ice melting? Who knows? Maybe polar bears will also do just fine on land, too…
Unfortunately, the reality is very different. It’s not just polar bears, walruses and the like that need the ice; we human beings will also directly and massively feel the consequences if it continues to disappear. After all, the sea ice is everyone’s business. Why? That’s exactly what we want to show you in this introductory section. We’ll explain just what sea ice is, how it forms, and its vital role in the Earth’s climate system.
Once you’ve read it, you’ll have a good initial overview. If you’d like to dig deeper into sea ice and learn more about e.g. the amazing diversity of species living in, on and below it, or how experts investigate the ice, we recommend checking out the detail section “Learn More”. There you’ll find out all about sea-ice physics, biology, measuring methods and modelling.
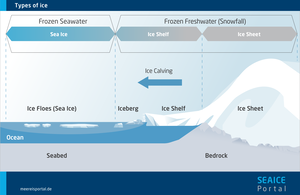
Sea ice refers to ice formed from frozen ocean water and which floats atop it. This distinguishes it from other types of ice like ice sheets, shelf ice and glacier ice, which are formed when precipitation freezes on land.
Ice, i.e., frozen water, can be found in several different forms on our planet. It forms glaciers in the mountains, massive ice sheets on Greenland and Antarctica, can be found in the permafrost soils of Siberia and Canada, and floats on the ocean as shelf ice or sea ice. Researchers refer to the totality of ice on Earth, in all of its forms, as the cryosphere.
Glaciers, ice sheets and shelf ice are made of frozen freshwater – i.e., of snow that has been compacted into ice over hundreds or even thousands of years. From the kilometre-thick ice sheets on Greenland and Antarctica, countless glaciers flow down to the coast, where in some cases they form ice shelves, which float on the water but remain connected to the land ice. Icebergs of various sizes frequently calve from the edges of these shelves and can drift for kilometres into warmer waters before they melt completely. In contrast, sea ice forms on the ocean itself and consists of frozen seawater. As such, unlike shelf ice it contains a certain amount of saline.
Sea ice is porous. Its crystalline ice matrix contains not only pockets of trapped air but also liquid, high-saline brine and solid salt complexes. Its gaseous, liquid and solid components make sea ice a complex, multiphase system, and one that is constantly changing thanks to ongoing freezing and melting processes.
Because of its crystalline structure and porosity, sea ice is less dense than seawater and always floats on its surface. Further, only 12 percent of the ice extends above the water as so-called “freeboard”; the remaining 88 percent lies below the surface.
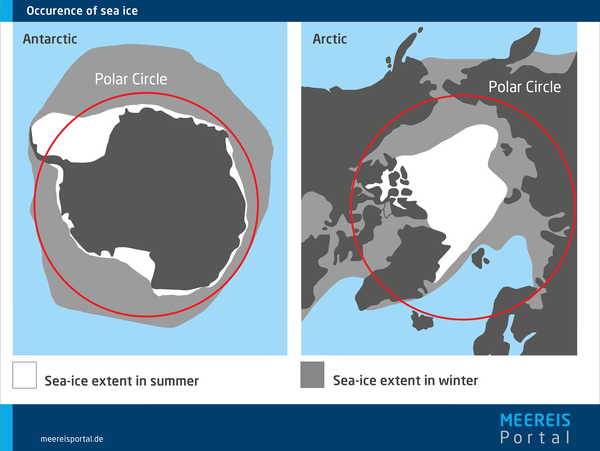
The largest concentrations of sea ice can be found in our planet’s polar regions – in the Arctic Ocean to the north and throughout the Antarctic to the south.
Sea ice can be seasonal – i.e., be formed and disappear again in the course of the seasons – or more permanent, surviving for years. Both types are chiefly found in the polar regions. Yet smaller areas of seasonal ice also form in other regions, like the Baltic Sea and the Sea of Okhotsk, which stretches from Russia to the coast of Japan.
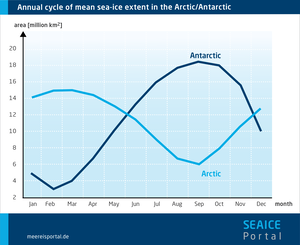
In the Arctic and Antarctic alike, the ice cover varies with the season. In the Northern Hemisphere, the seasonal ice melts in summer, reaching its minimum area in September – which means that, at this time of year, only multiyear ice still floats atop the Arctic Ocean. Afterwards, thanks to newly formed seasonal ice, the ice cover expands far into the adjacent seas of the Arctic Ocean, reaching its maximum extent in March. Here, the winter growth phase and summer shrinking phase are each six months long.
This works a bit differently in the Southern Hemisphere. The Southern Ocean surrounding Antarctica is home to extensive sea ice in winter, but virtually ice-free in summer; there is far less multiyear ice here than in the north. In addition, the winter growth phase, at seven months, is longer than the five-month summer phase. This greater seasonal variation is due in part to the fact that, unlike in the Arctic Ocean, sea-ice growth in the Antarctic is not limited by larger islands or continental landmasses.
Together with the continents’ varying snow cover, the seasonal change in sea-ice cover is one of the most remarkable changes on the surface of the Earth and clearly visible from space. Moreover, the sea-ice cover has a major influence on ocean currents, weather and our planet’s climate.
The ice on the ocean influences our planet’s energy budget, ocean currents, weather and climate. It also offers a habitat for various species and serves as a key early-warning system for climate change.
From a physical standpoint, the ocean’s surface is a boundary layer between the ocean and atmosphere, within which vertical exchanges of energy, inertia and mass take place. For example, a warm ocean warms the cold air above it – thermal energy is transferred from the ocean to the atmosphere. The ocean also helps determine how solar energy is distributed across the face of the planet – and therefore, what type of weather and climate is predominant in a given region.
Sea ice influences this energy flow in a number of ways.
- Like a giant, frozen lid, it reduces exchanges – of energy, for instance – between the ocean and atmosphere.
- It changes the aerodynamic roughness of the ocean’s surface, which affects how air masses move over it.
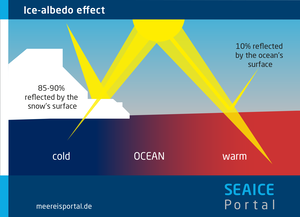
- It increases the ocean’s surface reflectivity (albedo) for sunlight. White sea ice reflects far more light back into space than does the much darker ocean.
- It has considerable influence on ocean currents, i.e., on how water masses within the ocean move across the planet.
Without the sea ice – which, depending on the season, covers an amazing three to seven percent of the Earth’s surface – the climatic conditions on our planet would be strikingly different.
As climate change progresses, the Arctic is warming far faster than the rest of the world. One of the main reasons for the difference is the ice-albedo feedback. In warmer climates, the bright sea ice melts, uncovering the darker ocean below. The latter begins absorbing the solar energy previously reflected back into space by the ice and grows even warmer. As a result, more sea ice melts and the entire process is amplified. Because the sea ice is so sensitive to rising temperatures, climatic changes can be recognised in the development of its extent even when there are still few signs in the rest of the world. Accordingly, in climate research the sea ice is an important indicator of – and a vital early-warning system for – climate change.
In addition to its importance for climatic processes, the sea ice offers a habitat for a diverse range of species – from tiny microorganisms to fish, polar bears and whales. These species live in, on or below the ice and are part of a highly complex, mutually independent and therefore fragile food chain. If just one link is lost, in some cases the entire chain can collapse.
First, smaller ice crystals form a soupy layer of ice on the ocean’s surface. Under calm weather conditions, it then becomes a continuous layer of ice (“nilas”); under more dynamic conditions, small pieces of ice (“pancake ice”) form instead.
If temperatures are low enough, ice crystals form in the topmost layer of the seawater; they look like small platelets or needles. These crystals rise to the ocean’s surface and gradually accumulate into a “soup” of sorts – known as “frazil ice”. When the weather conditions are calm, with no wave activity, this in turn forms a thin but unbroken layer of ice, referred to as “nilas”. At first, this crust of ice is so thin that it appears black, because the dark ocean below can be seen. Over time, nilas becomes thicker and lighter, with the surface remaining smooth and even.
In contrast, when conditions are less calm, with wind and waves, the frazil ice forms smaller, non-continuous sheets of ice that resemble pancakes (“pancake ice”). These tiny sheets collide, stack up, and ultimately freeze together, producing a sheet of ice that is also thick, but with an uneven surface.
Pancake ice
Under more dynamic weather conditions, the frazil ice condenses into a soupy layer measuring a few centimetres to one metre in thickness. Once wave activity has turned it into a uniform mixture (homogenisation), the result is referred to as “grease ice”.
If these weather conditions persist, the grease ice can form pancake-like pieces of ice. They have a diameter of up to five metres, can be up to 70 centimetres thick, and have thickened edges. These edges are produced by collisions with other floes and by seawater washing over them, leaving behind additional ice crystals.
Due to the choppy waters, no solid sheet of ice is formed; as a result, heat can still be directly transferred from the ocean to the atmosphere through the gaps between the “pancakes”. The water cools further, intensifying the pancakes’ growth. Because of the wave activity, the sheets of ice gradually pile on top of (“raft over”) one another. The soupy ice that washes over them acts like glue when the pieces subsequently freeze together. In this way, a sheet of ice is produced that is up to two metres thick, with an uneven surface but stable, and which nearly completely absorbs any wave activity. It also largely blocks thermal flows between the ocean and atmosphere and the sea ice’s growth rate declines sharply. On its underside, the ice continues to grow, albeit slowly. Here, column-shaped ice crystals (columnar ice) form, which can be up to ten centimetres long and clearly differ from the platelets and needles that make up frazil ice.




Nilas

When weather conditions are calm, the frazil ice forms a continuous sheet of new ice (“nilas”). If the nilas is less than five centimetres thick, the darker seawater below can be seen through it; this is referred to as “dark nilas”. The sheet of ice is still flexible and has a consistency similar to that of butter or ice cream.
As ice crystals gather on its underside, the nilas gradually grows further, becoming thicker, stronger and less transparent. It becomes lighter and lighter, transitioning from grey to white. As such, the surface brightness is a good indicator of its age.
Nilas can be up to two metres thick and has a smooth, even surface. Because it dampens thermal transfer between the ocean and atmosphere, the ice-formation rate now drops considerably. On its underside, long, column-shaped ice crystals (columnar ice) begin forming.
Ice that has freshly formed in a given winter is referred to as “seasonal ice”. If it survives one or more summer melting phases, it is dubbed multiyear ice.
Sea ice grows and melts in a seasonal cycle. In the Arctic (Northern Hemisphere) it grows in winter, at the same time it is melting in the Antarctic (Southern Hemisphere, where it’s summer). Then the process is reversed and the Arctic sea ice melts in summer, while growing again in the Antarctic. As such, the sea-ice cover alternates between two extremes in the north and south alike – the sea-ice minimum at the end of summer and the sea-ice maximum at the end of every winter.
Ice that has freshly formed in a given winter is referred to as “seasonal ice”. If it survives one summer melting phase, it is then called “second-year ice” because, by the second winter, it is in its second growth phase. If the same sea ice survives another summer, it is called “old ice”.
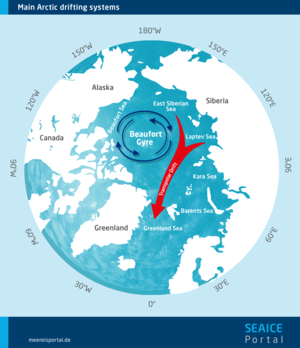
Multiyear ice is chiefly found in the Arctic Ocean. In the colder parts of the ocean near the North Pole, extended areas remain covered with old floes throughout the summer. If the weather conditions are calm, a layer of new ice (nilas) forms between them in the following winter. In the Beaufort Gyre, at the centre of the Arctic Ocean, old ice floes can endure for seven to ten years before being transported by the Transpolar Drift to the North Atlantic, where they ultimately melt.
Unlike the North Pole, the South Pole is not an ocean region; it is covered by the Antarctic landmass. Accordingly, the Antarctic Ocean surrounding the continent only begins comparatively far from the pole, between 50° and 70° S. In addition, it is choppier and stormier than the central Arctic Ocean. Thanks to the combination of these two factors, very little of the freshly formed ice in the Antarctic survives the summer; ocean currents only bring together the remaining multiyear ice in the Weddell Sea east of the Antarctic Peninsula and in the Ross Sea in the western Antarctic.
The sea-ice cover is rarely more than three metres thick. However, dynamic processes can produce stacks of broken-up ice that are up to 50 metres thick.
Because sea ice is characterised by low thermal conductivity, it has an insulating effect on the water layers below it. Consequently, the thicker the ice becomes, the more its freezing rate declines. At thicknesses of three metres or more, the melting processes are just as fast as the freezing processes. The ice is then in “equilibrium” and no longer becomes thicker.
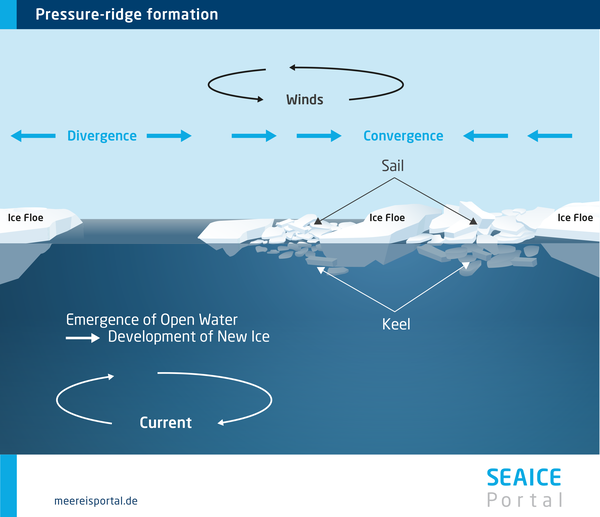
Nevertheless, dynamic processes can produce ice accumulations that are far thicker. The sheets of ice, measuring a few metres thick, are constantly in motion due to winds and ocean currents. As a result of this motion – as well as tides and long-wave swell – they break up, producing floes with channels of open water between them.
These floes are then transported across the ocean by winds and currents (sea-ice drift). In the Arctic, this mainly involves two major drift systems. In the Beaufort Gyre north of Alaska, the ice circulates clockwise. It is gradually fed into the Transpolar Drift, which transports ice from the coast of Siberia over the North Pole to Fram Strait between Svalbard and Greenland, where it then melts in the Greenland Sea.


When the pressure on the floes, exerted by the wind and water, is particularly high, the sheets can stack up on top of each other, forming hummocks with thicknesses of 50 metres or more. The high pressure and the soupy ice, which splashes onto the pack ice and acts like glue, bond it together and strengthen it.
In the Arctic, due to climate change the sea-ice cover has been declining dramatically for years. In contrast, it is still largely stable in the Antarctic.
The Arctic is especially hard-hit by climate change. For example, over the past 43 years, the mean air temperature in the High North has risen by more than 3 degrees Celsius – far more than anywhere else in the world (Rantanen et al., 2022). At the same time, the scientific community has observed a substantial decline in Arctic sea-ice cover. This becomes particularly apparent in a comparison of the annual data for September, the time of year when, following the summer melting, the Arctic sea ice reaches its lowest extent (September minimum), before rebounding again in the following winter.
Over time, the Arctic’s September sea-ice cover has dramatically decreased, by roughly 13 percent per decade: whereas roughly 7 million square kilometres of ocean were still covered with ice in the 1980s, today only 4.5 million square kilometres are. If this trend continues, before 2050 the Arctic will be nearly ice-free in some summers – with grave consequences for the entire ecosystem.
The decline in Arctic sea-ice cover and the rising temperatures produced by climate change are mutually amplifying. This is due to the ice-albedo feedback. Consequently, the September minimum in the Arctic is an important indicator of the progression of climate change.
In contrast, to date there is no clearly recognisable trend of declining sea-ice cover in the Antarctic. In the course of the annual cycle, the sea-ice extent regularly varies – and has done so fairly consistently for the past several decades – from 19 million square kilometres in winter to 3 million square kilometres in summer.
Rantanen, M., Karpechko, A.Y., Lipponen, A. et al. (2022): The Arctic has warmed nearly four times faster than the globe since 1979. Commun. Earth Environ. 3, 168, doi.org/10.1038/s43247-022-00498-3.
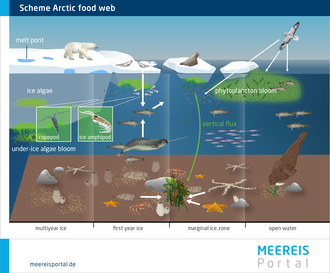
The up to 20-metre-wide, darker melt ponds allow a great deal of sunlight to penetrate the ice. As a result, it becomes warmer and melts more rapidly.

Over the past decade, the Arctic sea-ice cover has not only lost area; it has also become younger and thinner. Where there was once several-metre-thick multiyear ice, there are now predominantly seasonal floes that become riddled with melt ponds in summer. According to observational data, up to 40 percent of the ice’s surface can be covered by these ponds.
In the course of the summer, the snow cover on the sea ice melts completely – leaving behind small ponds of meltwater. In broad expanses of the Arctic, these ponds form in a matter of days, often in the first few weeks of June. They remain until September, when the surface starts refreezing. The majority of these freshwater ponds have a diameter of between 3 and 20 metres. Their colour depends on the thickness of the ice below them, which allows the dark (black) ocean to show through to a greater or lesser extent. Accordingly, they tend to be more turquoise on thicker multiyear ice and dark blue to black on thinner seasonal ice.
The ponds have significant effects on the ice. White sea ice and snow reflect nearly all incoming sunlight. In contrast, the dark melt ponds reflect only a fraction of the light, allowing more of it to penetrate the sea ice and – through the ice – the ocean.
As a result, young and thin ice with numerous melt ponds allows roughly three times as much light through as does older ice. Further, it absorbs twice as much sunlight. Consequently, the ice warms more quickly and melting is intensified – the ice effectively melts “from within”.
Melt ponds also affect satellite-based readings. The satellites’ sensors mistake them for patches of open water, instead of sea ice. This can lead to considerable inaccuracies.
Despite the extreme conditions, an amazing biodiversity has developed. The polar food web is highly complex and ranges from tiny algae to polar bears, penguins and whales.
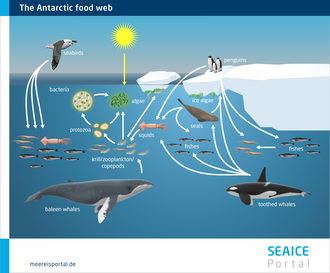
At first glance, the Arctic and Antarctic sea ice may seem harsh and inhospitable. But guess again: especially below the ice, a diverse range of highly specialised marine organisms have managed to defy the challenging conditions. From tiny single-celled organisms to gigantic blue whales, they form a complex food web in which various predator-and-prey relationships mean the survival of one depends on the wellbeing of the other.
At the bottom of the food pyramid (level 1) are the primary producers – countless microalgae and other microorganisms that use photosynthesis. They live on, within, and (particularly) on the underside of the ice. Here they use the sunlight that penetrates the ice to produce biomass. The biomass in turn is the central basis of nutrition for all other organisms.
At level 2 of the pyramid we find the smallest organisms – including copepods and krill – which live in the ocean and feed on the microalgae below the ice. At level 3, these organisms are eaten by fish larvae and juvenile fish, which in turn become prey for seals, birds, whales and polar bears. Of course, the reality is far more complicated than this simplified model. Nevertheless, the pyramid clearly shows how every species – no matter how small – has its own unique and essential role in the great symphony of sea-ice organisms. And that, if it were to be lost, it would have consequences for all polar species.
With each step up in the pyramid, a large percentage of the energy stored in biomass is lost. For example, when a seal eats fish, the meat it eats doesn’t turn into body mass at a 1-to-1 ratio. Instead, the seal “burns” a large fraction of the energy to produce body heat, to move, and for other functions. As such, only a small percentage is used to create its own tissues. This is why no food pyramid can go on forever; at the peak there are only a handful of “top predators”, like the polar bear, and the biomass energy they receive is just enough for their survival.
The only way to survive in the ice is to make special adaptations to the extreme conditions. In this regard, protection from the cold and frost is at the top of the evolutionary priority list. Nearly all larger animals use the “onion principle” – including the polar superstars: penguins in the Antarctic and polar bears in the Arctic. To keep out the cold, penguins use a centimetre-thick layer of fat and several layers of densely packed feathers, which are even waterproof, equipping them for diving in the frigid waters of the Southern Ocean. Arctic polar bears also have a thick fatty layer and special, oily and water-resistant coat of fur. Atop their dense undercoat lies their outer coat; its hollow hairs, together with the inner layers of the “onion”, provide excellent insulation. Another, amazing example: the “antifreeze” glycoproteins produced by some polar fishes, which keep their bodily fluids from freezing.
If you’d like to learn more about this biological symphony and the most important sea-ice organisms, we recommend visiting the “Sea-ice Biology” section.
Neil A. Campbell, Lisa A. Urry, Michael L. Cain, Steven A. Wasserman, Peter V. Minorsky, 2017, Author Biology: A Global Approach, Global Edition (English Edition) 11. Auflage, Chapter: The Ecology of Life; Seite 1280, Figure: 54.15 An Antarctic marine food web.
Snow, Water, ice and Permafrost in the Arctic (SWIPA) (2011); climate change and the cryosphere, Arctic Monitoring and Assessment Programme, Oslo, S. 9-33, Grafik 9.29

Glossary
Science invariably involves terminology. To help you find your way, we’ve linked key terms to an extensive glossary. Just click on the term within the text and you’ll find a brief definition – from A for albedo to Z for zooplankton.